Mission
Our mission is to train the next generation of leaders in Medical Biochemical Genetics, equipping them with the knowledge and skills needed to provide exceptional patient care, conduct innovative research, and advance the field of Biochemical Genetics. Through a rigorous and comprehensive Medical Biochemical Genetics Fellowship Program, we strive to provide our fellows with a deep understanding of the diagnosis, treatment, and management of patients with inborn errors of metabolism.
Program Overview
The University of Washington School of Medicine’s Medical Biochemical Genetics Fellowship is based at Seattle Children’s Hospital and provides care to children and adults at Seattle Children’s Hospital and the University of Washington. Our program covers a diverse population from a large region covering five states in the Pacific Northwest (i.e., Washington, Wyoming, Alaska, Montana and Idaho), and newborn screening for Hawaii. Fellows will participate in clinical care and learn from patients from all areas of inborn errors of metabolism in clinic, and act as a member of the primary admitting in-patient service and hospital consultation service. Fellows will also have opportunities to participate in the vast clinical and research programs at both institutions and in the clinical biochemical genetics laboratory to learn the complex interpretation of biochemical genetic testing.
During their fellowship year, fellows have an intensive exposure to inpatient and outpatient biochemical genetics and learn the management of a wide variety of acute and chronic inborn errors of metabolism. These principles of treatment include dietary management, medications, enzyme replacement therapy, and solid organ or bone marrow transplantation. Through the year, fellows will have the opportunity to rotate in the Biochemical Genetics Laboratory. Fellows are encouraged and supported to present their research at national and international meetings. This training focuses on providing fellows the skills necessary to become independent medical biochemical geneticists.
Departmental Programs
Seattle Children’s Biochemical Genetics Clinic
Biochemical genetics involves diagnosing and treating metabolic diseases. These are problems with how the body makes, breaks down or uses proteins, fats or carbohydrates. They are caused by genes that are not normal and cannot make the proteins the body needs. (Proteins are found in the blood, organs, saliva and other parts of the body. They are important to all bodily functions.)
These diseases are called inborn errors of metabolism. Our team helps to diagnose, monitor, and manage these diseases. We work closely with doctors in other departments at Seattle Children’s and in our Genetics Lab. Most of our patients have complex health problems. They need coordinated, long-term care from a team of experts, like ours.
Diagnosing and treating metabolic diseases early can improve a child’s quality of life. The Washington State Department of Health screens newborns for these diseases. We work with them to provide any follow-up genetic testing and care that newborns need.
Our program is part of the American Board of Medical Genetics–certified training program at the University of Washington. Another name for this area of genetics is metabolic genetics.
Phenylketonuria (PKU) Program at UW
The Cristine M. Trahms Program for Phenylketonuria at the University of Washington is made up of a team of experienced health care professionals. Together, this team provides services to people with PKU and their families in the state of Washington. The program is primarily funded by the Washington State Newborn Screening Program. The State Genetics Program provides families with subsidized formula (paid for by their medical insurance). The programs are closely coordinated to ensure follow-up and appropriate management of individuals with PKU. The PKU program provides multidisciplinary care for patients and families inherited with the PKU disorder. The clinic offers an all-encompassing approach to care providing medical and nutritional assessments as well as nutrition education and developmental testing for all children in the PKU clinic. Our goal is to provide individualized care to each family with our goal of family support and nutritional education to be built into each clinic visit and beyond.
The Urea Cycle Disorders (UCD) Program at Seattle Children's
The UCD Program provides multidisciplinary care including consultation, examination, testing, diagnosis, treatment, and genetic counseling for patients with urea cycle disorders and their families. Our goal is to provide patients and families with a comprehensive medical home that is centered on the patient and family. Our mission is to provide each patient with the knowledge, ability, and proper tools to allow them to manage their disorder. The biochemical genetics team is the core within a larger program dedicated to the management of these diseases and providing care by UWMC and SCH specialists in many areas of medicine including psychology, neurodevelopment, neurology, gastroenterology, and organ transplant. The clinic team follows a large group of children and adult patients across the spectrum of UCDs. Our group also works with local hospitals to coordinate the treatment of care during acute emergencies and in routine follow-up closer to each patient's home.
We are active members of the Urea Cycle Disorders Consortium Longitudinal study, sponsored by the Rare Disease Clinical Research Network at the NIH along with the National Urea Cycle Disorders Foundation in order further improve treatment, quality of life, and our understanding of UCDs and to implement newborn screening for all UCDs – including ornithine transcarbamylase deficiency. We are also actively involved in multiple collaborations with urea cycle disorder support groups and with clinical research trials developing novel treatments of UCDs.
Newborn Screening Program
 Since Washington State’s Newborn Screening Program began in 1963 with PKU, the Biochemical
Since Washington State’s Newborn Screening Program began in 1963 with PKU, the Biochemical
Genetics Program has been providing confirmatory diagnostic testing, clinical treatment and care coordination for babies identified with metabolic diseases from infancy to adulthood. The Biochemical and Molecular Genetics Laboratory staff works very hard to prioritize and analyze these samples as quickly as possible. Currently Washington State is screening for 6 amino acid disorders, 5 fatty acid oxidation disorders, 7 organic acid disorders, Pompe disease, X-linked adrenoleukodystrophy, MPS I, and several other congenital disorders. Details of each disorder can be found at www.doh.wa.gov/nbs

The Washington State Department of Health (DOH) continues to provide the Newborn Screening confirmation & follow-up program possible with contract funding for the consultative services and additional testing required to confirm or rule out presumptive diagnoses. This confirmatory diagnosis allows for the earliest possible treatment to begin, giving these children the best quality of life and in some cases a normal or nearly normal lifestyle that would not have been possible without this early care. DOH also continues to provide contract funding for metabolic dietitian services for the Newborn Screening Follow-up Program, as well as for outreach services for semi-annual outreach at the Biochemical Genetics Clinics in Spokane.We continue to be actively involved with the Western States Genetic Services Collaborativeto study the outcomes of infants with abnormal newborns screens and, in particular, the long-term outcomes of infants identified with Very Long-Chain Acyl-CoA Dehydrogenase (VLCAD) Deficiency.
The Lysosomal Storage Disease (LSD) Program at UW/SCH
The LSD program provides multidisciplinary care including consultation, examination, testing/ diagnosis, treatment, monitoring, and genetic counseling for patients with lysosomal storage diseases and their families. The patients' medical home is centered with the Biochemical Genetics team. The program is dedicated to the management of these rare diseases and provides care by UWMC and SCH specialists in many areas of medicine including ophthalmology, orthopedics, cardiology, neurology, nephrology, pulmonary, otolaryngology, neurodevelopmental, radiology and anesthesia. Our group also works with local hospitals to facilitate transition of treatment to centers closer to each patient's home or for infusions to be done in the patient’s home. Long-term follow-up of patients with these rare diseases is essential.
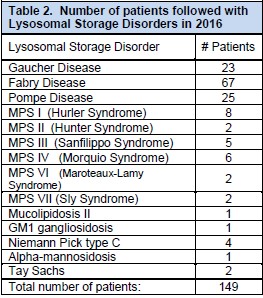
Patient education and support meetings are held periodically, providing patients and their families an opportunity to mingle with other families and hear about recent advances in the field. There is much ongoing research in the field of lysosomal storage disorders. The UWMC and SCH teams currently have active patient registries for MPS I, MPS II, MPS IV, MPS VI, Pompe disease, Fabry disease, and Gaucher disease. The clinic team follows a large cohort of patients across the spectrum of LSDs as illustrated in Table 2 and long term follow-up of patients with these rare diseases is essential as is continuing to keep abreast of new developments in therapies and care. A clinical trial for Hunter syndrome is in progress at SCH under IRB approval to evaluate long term safety and clinical outcomes of intrathecal Idursulfase enzyme replacement. Another clinical trial on patients with Pompe disease to evaluate the efficacy and safety of alglucosidase alfa produced at the 4000L scale was successful and the medication was approved by the FDA.
Laboratory/Research
Biochemical Genetics Laboratory
The Biochemical Genetics Laboratories (SCH) are led by Dr. Sihoun Hahn and Dr. Anna Scott. The Biochemical and Molecular Genetics Laboratories serve as a key component of the Biochemical Genetics Program. The laboratories provide rapid, comprehensive diagnostic and monitoring results along with guidance for follow-up. Our laboratory is committed to high quality results. To ensure accuracy, the laboratory is participating in a variety of external proficiency testing programs offered by various national and international agencies (ERNDIM; European network, CAP; College of American Pathologists, CDC; Center for Disease Control).
Research and Development (R & D) Laboratory
The Biochemical Genetics program’s CLIA-certified R&D Laboratory is located at the Seattle Children’s Research Institute (SCRI): Center for Integrative Brain Research (Center Director. Dr. Jan-Marino Ramirez). The facility is fully equipped with a LCMS/ MS and an Illumina Genome Analyzer IIx. Since 2007, the R&D team has continuously focused their efforts on refining current methodologies and developing new tests, with a fundamental goal to improve clinical practice through the implementation and integration of routine laboratory testing. In particular, the lab aims to develop and validate clinical tests to diagnose various metabolic and genetic disorders by utilizing tandem mass spectrometry.
The R&D team has a long-standing interest in developing a mass spectrometry based assay for diseases characterized by the absence and/or reduction of protein markers. We use a highly sensitive and targeted analytical technology, selected reaction monitoring mass spectrometry (SRM-MS), to measure peptides of our interest, which enables efficient translation of protein markers into clinical use as screening and diagnostic tests. As proof-of-concept, we previously demonstrated that SRM-MS analysis of signature peptides can correctly identify patients lacking specific protein markers of three life-threatening Primary Immunodeficiency Disorders (Proteomics Clin Appl, 2012, R21AI85488). Our efforts (R56AI106784, R21HD069890, and CRFS- 2015-004) to improve the sensitivity of our assay by combining SRM-MS with antibody-based enrichment of target peptides (immuno-SRM) showed that the method can detect extremely low abundance marker proteins of congenital disorders such asCD3e (for SCID), BTK (for XLA), WASP (for WAS), CTNS (for Cystinosis), and ATP7B (for Wilson disease) in dried blood spots. In 2016, we published the proof-of-concept study demonstrating that the immuno-SRM assay readily distinguishes affected cases of Wilson disease from normal controls (p<0.0001) (Journal of proteome research, 2016). We also received an NIH grant (R01AI123135) to further study and develop a multiplexed immuno-SRM assay for screening11different primary immunodeficiency disorders. We believe that our proteomics based approach will provide an efficient and inexpensive screening for a
broad range of genetic disorders. At the heart of translational research, the R&D Laboratory’s goal is to bridge basic research and the clinical applications of novel tests developed for effective patient diagnosis and treatment.

Helpful Links
Informational Videos
- Introduction by the Vice Dean of Graduate Medical Education
- Your Life Outside of Work
- Your Life at Work
- Research at Seattle Children’s
- Message from Dr. Walker, Department of Pediatrics Chair
Programs
- UW Pediatric/Genetic Medicine Division
- UW Clinical Genetics Residency
- Seattle Children’s Genetics
- Seattle Children’s Biochemical Genetics
- Seattle Children’s Hospital
Associations
Curriculum Goals
The goal of the medical biochemical genetics experience is to learn about the diagnosis, genetic counseling, and management of patients with the full range of inborn errors of metabolism. The focus is on clinical care and management, and fellows will hone their skills at recognizing clinical findings and patterns of clinical and laboratory findings that point to inborn errors of metabolism. Further, they will become proficient at recognizing early warning signs of change in clinical status in patients with established diagnoses so that more severe metabolic decompensation can be avoided. As part of this experience, fellows will learn the principles that underlie common analytical procedures such as amino acid, organic acid, and acylcarnitine analyses both by observation in the laboratory and by regular involvement with the interpretation process throughout the fellowship training. During this fellowship the resident will develop the necessary knowledge base and skills to interpret and confirm newborn screening tests, accurately and compassionately counsel families during the process of confirmatory testing, and provide high quality management of those disorders. The fellow will be an integral part of our Metabolic Service that provides hospital care, consultation and outpatient care to individuals with inborn errors of metabolism.
Curriculum Summary
The fellowship program is located at Seattle Children’s Hospital (SCH), along with the Biochemical Genetics Laboratory (BGL) at SCH, and the University of Washington Medical Center with the support of the Department of Pediatrics at the University of Washington School of Medicine. The “Biochemical Genetics Service” provides inpatient care and consultation, outpatient care and consultation, and telephone consultative services to physicians in Washington State and the WWAMI Region (Washington, Wyoming, Alaska, Montana, and Idaho) and other areas in the Pacific Northwest and around the nation. Fellows spend one full year in clinical (patient-care oriented) activities, of which one full month will be spent in the BGL laboratory becoming familiar with the analytical methods used and developing an understanding of the implications of methods and analytical processes on clinical interpretation and application. The remaining training time will be spent caring for individuals with suspected or documented inborn errors of metabolism, both inpatients and outpatients. During that time the trainee is expected to be involved in academic activities, including teaching students and residents, preparing and presenting formal presentations, preparing a publication, and taking part in the clinical research activities of the Biochemical Genetics Service.
Curriculum Scheduling
Year 1 Requirements:
| Activity | # of participating months | Day of week | Frequency/month | ½-day or full day |
|---|---|---|---|---|
| BCG Clinic | 11 | Tue, Thurs, Fri. | 7 – 14 | 1/2 |
| IHDD/LSD | 11 | 1st Tuesday | 1 | 1/2 |
| IHDD/PKU | 11 | 3rd Wednesday | 1 | Full |
| IHDD Metabolic | 10 | 1st Thursday | 1 | 1/2 |
| BCG Laboratory* | 1 | Mon – Fri | 23 | Full |
Additionally, fellows will have weekly time dedicated to learning/teaching, education, and research as well as the option to pursue Elective rotations throughout the hospital.
*Details Regarding BCG Laboratory Rotation:
- Be present in the labs as much as possible between 9:00 a.m. and 5:00 p.m. during the rotation.
- Review cases that are ready for sign-out, including the indication for testing, any medical records available through EPIC for internal patients, and test results.
- Participate in daily sign-out of cases.
- Observe lab scientists and technologists to learn how the methodologies are implemented.
Curriculum Objectives
Inborn Errors of Metabolism
- Evaluation and diagnosis of patients with suspected inborn errors of metabolism
- Developing and applying a specific diagnostic and management plan for individuals with abnormal newborn screens ordering appropriate tests for the clinical situation
- Accurately interpreting biochemical screening and diagnostic test results, with awareness of important artifacts and sources of metabolites
- Acute and chronic medical and nutritional care of individuals with IEMs
- Effectively communicating recommendations and follow-up plans to patients, families and referring physicians
- Working with, and when appropriate leading, teams of healthcare providers in the management of acutely ill children with IEMs
- Prescribing and managing enzyme replacement therapy for IEMs
- Skin biopsy for fibroblast culture and diagnostic testing
Phenylketonuria Unit
- Evaluation and diagnosis of patients with suspected Phenylketonuria
- Developing and applying a specific diagnostic and management plan for individuals with abnormal newborn screens ordering appropriate tests for the clinical situation
- Accurately interpreting biochemical screening and diagnostic test results, with awareness of important artifacts and sources of metabolites
- Acute and chronic medical and nutritional care of individuals with Phenylketonuria
- Effectively communicating recommendations and follow-up plans to patients, families and referring physicians
- Working with, and when appropriate leading, teams of healthcare providers in the management of acutely ill children with Phenylketonuria
- Prescribing and managing enzyme replacement therapy for Phenylketonuria
- Skin biopsy for fibroblast culture and diagnostic testing
Lysosomal Disorders
- Evaluation and diagnosis of patients with suspected Lysosomal Disorders
- Developing and applying a specific diagnostic and management plan for individuals with abnormal newborn screens ordering appropriate tests for the clinical situation
- Accurately interpreting biochemical screening and diagnostic test results, with awareness of important artifacts and sources of metabolites
- Acute and chronic medical and nutritional care of individuals with Lysosomal Disorders
- Effectively communicating recommendations and follow-up plans to patients, families and referring physicians
- Working with, and when appropriate leading, teams of healthcare providers in the management of acutely ill children with Lysosomal Disorders
- Prescribing and managing enzyme replacement therapy for Lysosomal Disorders
- Skin biopsy for fibroblast culture and diagnostic testing
Biochemical Genetics Laboratory
- Develop life-long self-learning skills to complement the body of knowledge acquired during formal training in clinical biochemical laboratory science and to develop analytical thinking skills and investigative techniques.
- Understand operation of biochemical genetics laboratory, including typical turnaround times.
- Recognize appropriateness, limitations and costs of major diagnostic assays performed.
- Understand major approaches for screening biochemical disorders: urine organic acid analysis (GC-MS), acylcarnitine profile (MS/MS), and amino acid quantification (HPLC).
- Recognize importance of CAP accreditation and prevalence of laboratory developed testing within the field of biochemical genetics working with Laboratory Supervisor.
- Learn about newborn screening and resources to identify state specific newborn screening information.
- Review a variety of cases to appreciate the biochemical spectrum of different inborn errors of metabolism as well as differences between individuals with the same diagnosis.
Elective Curriculum Objectives (Optional, based on prior training)
NICU
During the NICU elective rotation, the Medical Biochemical Genetics Fellow should have the following goals and objectives as they relate to the care of a child with an IEM:
- To observe and become familiar with medical care of the critically ill newborn, including principles of medication administration, access, nutrition, and airway support.
- To develop a knowledge base and resource base for future learning in neonatal-perinatal medicine.
- To learn to recognize unique disorders and co-morbidities in the prenatal infant that may influence the care of the newborn with an IEM.
- Understand important patient and family care issues that may impact the newborn as they relate to the newborn with an IEM.
- Develop at least one topic or hypothesis where care of the infant with an IEM may be improved when they are admitted to the NICU.
- Present an IEM topic in an education session for the NICU care team and residents.
PICU
During the PICU elective rotation, the Medical Biochemical Genetics Fellow should have the following goals and objectives as they relate to the care of a child with an IEM:
- To observe and become familiar with medical care of the critically ill child, including principles of medication administration, access, nutrition, and airway support.
- To develop a knowledge base and resource base for future learning in pediatric critical care medicine.
- To learn to recognize unique disorders and co-morbidities in the prenatal infant that may influence the care of the child with an IEM.
- Understand important patient and family care issues that may impact the child as they relate to the child with an IEM.
- Develop at least one topic or hypothesis where care of the child with an IEM may be improved when they are admitted to the PICU.
- Present an IEM topic in an education session for the PICU care team and residents.
Prerequisites
Fellows must have graduated from an accredited medical school and have completed a residency program at an accredited institution. The individual must be board eligible/certified in Medical Genetics and Genomics.
Applications
To be considered, please send in email to our program coordinator Jackie.Foss@seattlechildrens.org:
- your CV
- a personal statement describing your interest in medical biochemical genetics
- 3 letters of recommendation
- USMLE step scores
- your future career plans
The Medical Biochemical Genetics Fellowship accepts US Citizens, J-1 or H-1B visas.
Application Deadline
Email our program coordinator Jackie for application deadline dates at Jackie.foss@seattlechildrens.org .
Interview Start and End Dates
Yearly from August 1 – January 15
The UW School of Medicine is committed to building and sustaining a multicultural community that fosters equity, diversity and inclusion. Our objective is to create a community that encourages participation and connection, and that values each individual's unique contribution, regardless of socioeconomic status, race, ethnicity, language, nationality, sex, sexual orientation, gender identity and expression, spiritual practice, geography, mental and physical status and age.
Seattle Children's Hospital

Seattle Children's Hospital is both a community hospital for greater Seattle and the pediatric referral center for the Northwest providing excellent pediatric care to meet the medical, surgical and developmental needs of children in the WWAMI region (Washington, Wyoming, Alaska, Montana, and Idaho). Serving as the main clinical training site for pediatric residents, this 423-bed hospital is conveniently located 1.5 miles from the University of Washington campus in a residential neighborhood of Seattle. The staff consists of University faculty and Seattle Children's full-time physicians.
Additional Information
UW Medical Center - Montlake (UWMC)

UWMC is consistently ranked among the nation's best general hospitals. As the only comprehensive clinical, research and learning health system in the five-state WWAMI (Washington, Wyoming, Alaska, Montana, Idaho) region, UW Medicine provides a higher degree of healthcare, ranging from primary and preventive care to the most highly specialized care for the most complex medical conditions.
From first of their kind, life-saving surgical procedures to routine adult, maternal and newborn medicine, we're training the next generation of medical professionals. By using the latest advances in medical technology and patient- and family-centered care, we're building a better future for our community.
University of Washington Institute on Human Development and Disability (IHDD)

IHDD is one of the nation's largest and most comprehensive interdisciplinary research and training centers focusing on a wide array of developmental disabilities. The Clinical Training Unit (CTU) at IHDD is an interdisciplinary program that provides training, research, and exemplary services in the assessment and treatment of children with or at risk for developmental disabilities, using a family-centered, community-based, culturally competent approach in a variety of clinical formats.
The Child Development Clinic provides diagnosis, assessment and management plans for children from early childhood to adolescence. The interdisciplinary teams include professionals from audiology, developmental/behavioral pediatrics, nursing, nutrition, psychology, occupational therapy, physical therapy, social work, and speech and language pathology to evaluate each child and make recommendations for care. Trainees participate in hands-on and didactic training which includes assessments, parent conferences, lectures, and report writing. Over 200 children are served per year with a wide range of diagnoses including intellectual disability, autism spectrum disorders, motor disabilities, learning disabilities, behavioral disorders, communication disorder, and attention-deficit hyperactivity disorder.
The High Risk Infant Follow-Up (HRIF) clinic at IHDD provides developmental follow-up of children from birth to age eight years. It provides an ideal setting for interdisciplinary training. The goal of the clinic is to provide early identification and referral for early intervention for the neurodevelopmental and neurobehavioral problems associated with prematurity, drug exposure and other biomedical and environmental risk factors for over 400 clients per year.
Seattle Children's Research Institute (SCRI)

As one of the nation's top five pediatric research centers, Seattle Children's Research Institute is dedicated to providing hope, care and cures to help every child live the healthiest and most fulfilling life possible. With one million square feet of clinical, laboratory and office space, a workforce of over 2,400 people and over $253 million in total extramural funding for the 2023 fiscal year, we are working toward the next wave of lifesaving discoveries.
The research institute is organized into centers, specializing in areas that include immunotherapy, tissue and immune transplantation, outcomes research, clinical and translational research, and child health and behavior. Researchers work in close collaboration with one another, their colleagues at partner institutions including the University of Washington and Fred Hutch Cancer Center and our healthcare providers at Seattle Children's Hospital. This collaboration allows our faculty to draw on a variety of disciplines and techniques as they pursue solutions to some of medicine's most complex problems.
Fellowship Leadership
Christina Lam, MD
Associate Professor, Division of Genetic Medicine Medical Director, Biochemical Genetics
Additional Participating Providers
Biochemical Genetics Laboratory, Seattle Children’s:
- Anna Scott, PhD, Director Clinical Biochemical Genetics Laboratory
Genetic Counselors:
- Teresa Campbell, MS, CGC, PhD
- Erika Beckman, MS, CGC
- Maria Mills, MS, CGC
- Kaitlin (Kait) Miller, MS, CGC
Genetic Counselor Assistant:
- Vivian Huynh
- Brenda Chavez Reyes
Dietitians:
- Erin Phelps, MS, CD, RDN
- Sarah Bell, MS, CD, RDN
- Holly Stevens, RDN, CD, RDN
- Beth Ogata, MS, RDN, CSP
- Mari Mazon, MS, CD, RDN
- Maryann Ruiz, MS, CD, RDN
Social Work:
- Gailon Wixson, MSW, LSWAIC
Nursing:
- Olivia Moore, RN, BSN, MBA
- Kasea Hildebrandt, RN, BSN
- Bernice Cristobal, RN, BSN
- Jeff Posadas, RN, BSN
- Angela Wipfler, RN, BSN
Physician Assistant:
- Maria Wright, PA-C
Clinical Research Coordinators:
- Hayden Vreugdenhil, BS
- Seishu Horikoshi, MBA
- Phi Duong, MS
Recent Alumni
Monica Penon Portmann, MD (2022-2023)
Seattle Children's/University of Washington
Sheri A. Poskanzer, MD (2019-2020)
St Luke's Boise
Irene Chang, MD (2018-2019)
University of California, San Francisco
Christina Lam, MD
Associate Professor, Division of Genetic Medicine Medical Director, Biochemical Genetics
Seattle Children’s Hospital
PO BOX 5371
M/O OC.6.820
Seattle, WA 98145-5005








